
The acceleration of an object depends directly upon the net force acting upon the object, and inversely upon the mass of the object. The second law states that the acceleration of an object is dependent upon two variables - the net force acting upon the object and the mass of the object. Newton's second law of motion pertains to the behavior of objects for which all existing forces are not balanced. The presence of an unbalanced force will accelerate an object - changing its speed, its direction, or both its speed and direction. According to Newton, an object will only accelerate if there is a net or unbalanced force acting upon it. Objects at equilibrium (the condition in which all forces balance) will not accelerate. The first law - sometimes referred to as the law of inertia - states that if the forces acting upon an object are balanced, then the acceleration of that object will be 0 m/s/s. Thus, the balanced methane equation above can also be interpreted as reading, "one mole of methane reacts with two moles of oxygen to produce one mole of carbon dioxide and two moles of water.Newton's first law of motion predicts the behavior of objects for which all existing forces are balanced. Because the mole refers to a standard number of atoms (or molecules), the term can simply be substituted into chemical equations. Chemical equations are discussed in relation to the number of moles of reactants and products used or produced (see our The Mole module). This is because single atoms (and molecules) are so tiny that they are difficult to isolate. In this example, methane is called theĪlthough we have discussed balancing equations in terms of numbers of atoms and molecules, keep in mind that we never talk about a single atom (or molecule) when we use chemical equations. Side of the equation to balance it and show that the excess oxygen is notĬonsumed during the reaction. In the example seen above, 3O 2 had to be added to the right 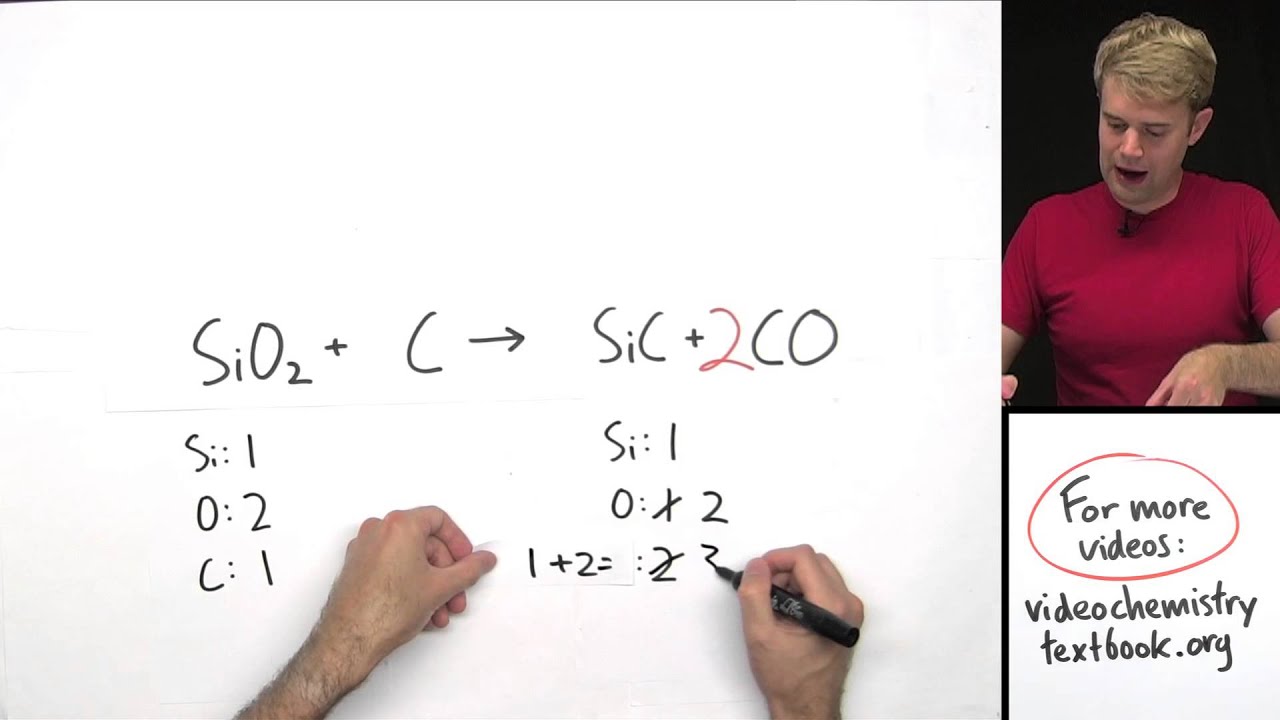
The unbalanced chemical equation would be written: During this reaction, chemical bonds break and re-form and the products that are produced are carbon dioxide and water vapor (and, of course, light and heat that you see as the flame). When you light the stove, you are supplying the activation energy to start the reaction of methane with oxygen in the air. Methane (CH 4) is a molecule that contains four hydrogen atoms bonded to one carbon atom. If you use a gas stove to cook your dinner, chances are that your stove burns natural gas, which is primarily methane. In order to write a correct chemical equation, we must balance all of the atoms on the left side of the reaction with the atoms on the right side. If no coefficient appears in front of a molecule, we interpret this as meaning one. In writing chemical equations, the number in front of the molecule's symbol (called a coefficient) indicates the number of molecules participating in the reaction. Given this information, one might guess that the equation for this reaction is written: To write the chemical equation for this reaction, we would place the substances reacting (the reactants) on the left side of an equation with an arrow pointing to the substances being formed on the right side of the equation (the products). If we had a container of hydrogen gas and burned this in the presence of oxygen, the two gases would react together, releasing energy, to form water. Let's take the reaction of hydrogen with oxygen to form water as an example. A chemical equation is shorthand that scientists use to describe a chemical reaction. A chemical reaction is the process by which substances bond together (or break bonds) and, in doing so, either release or consume energy (see our Chemical Reactions module). To view it, go here.Ĭhemical reactions happen all around us: when we light a match, start a car, eat dinner, or walk the dog. An updated version of the Chemical Equations module is available.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
